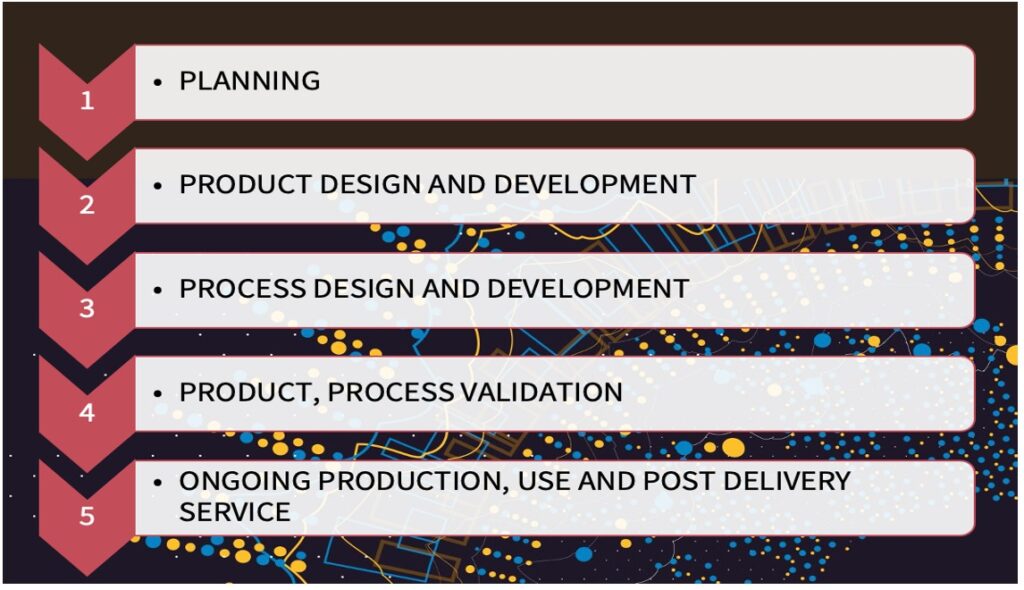
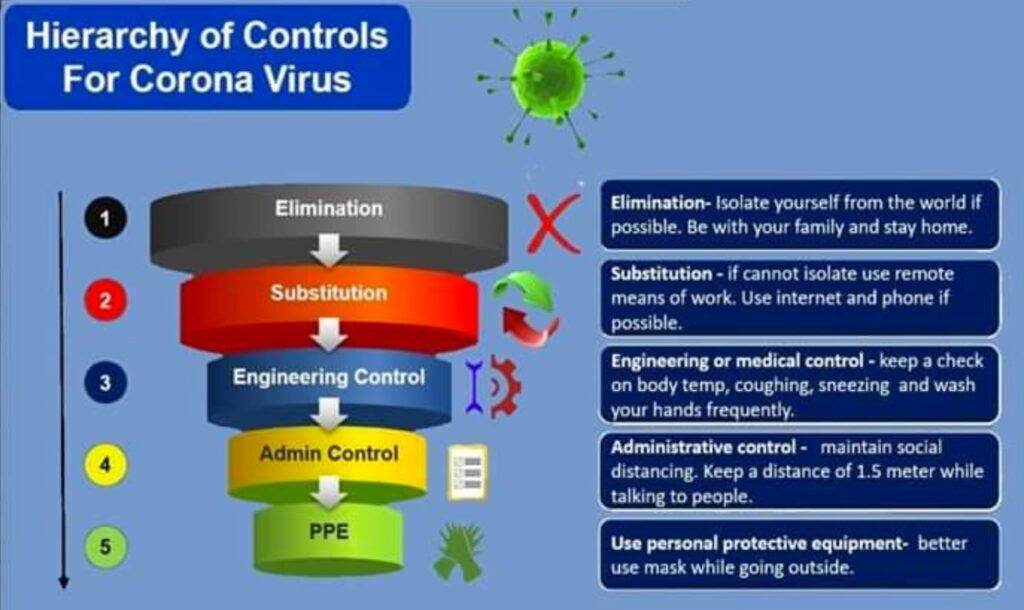
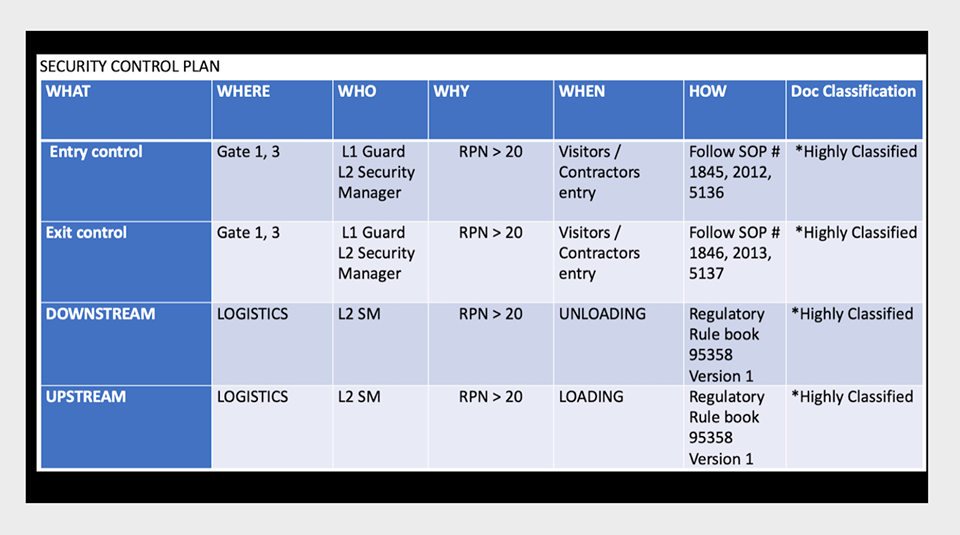
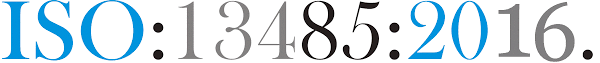LeadSOC Acknowledges Global Quality Services for ISO 9001, ISO 27001, and TISAX Certification Support
LeadSOC recognises Global Quality Services for guiding its quality and information security certification programs! LeadSOC Technologies Pvt. Ltd. has announced […]
ISO 17025: Building Global Laboratory Excellence – A Perspective from AlUla, Saudi Arabia
In today’s knowledge-driven world, laboratories play a central role in ensuring the safety, accuracy, and reliability of products and services […]
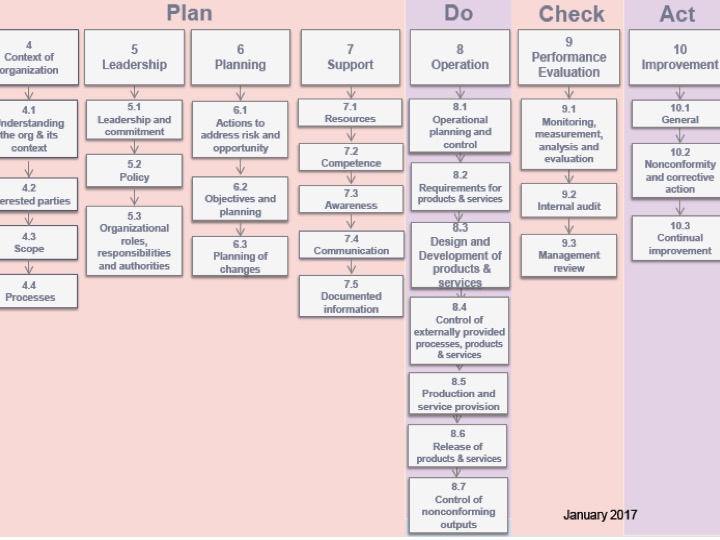
ISO 9001 Certification
Businesses across every industry need to deliver consistent quality, meet customer expectations, and comply with regulatory standards. To achieve this, […]
Grow Your Medical Devices Business in Saudi Arabia with ISO 13485 Certification
The medical devices industry is one of the fastest-growing sectors in Saudi Arabia, driven by the Kingdom’s Vision 2030 initiatives […]
From Compliance to Confidence: Implementing ISO 18788 (Security Management)
As Saudi Arabia accelerates its Vision 2030 initiative to diversify the economy, strengthen culture, and build inclusive communities, effective security […]
Building Trust in Aerospace: The Importance of AS9100 Certification
The aerospace industry is one of the most complex and highly regulated sectors in the world. Whether it’s designing commercial […]
Create a trusted brand assurance platform for food industry: FSSC 22000 Certification
Food is the first of the basic needs and a primary source of energy and existence. We relish it in […]
Validation and verification of Greenhouse Gas (GHG) using ISO 14064 certification
“Voluntarily monitor the emission of GHG – The earliest you do, the more safer you can be” ISO 14064 – […]
Navigating the Skies: A Deep Dive into AS 9100 Certification
In the rigorous aerospace industry, quality is not merely a choice—it is a must. AS 9100 accreditations, a widely accepted […]
ISO 39001: Paving the Way for Safer Roads
In Saudi Arabia’s crowded cities and broad highways, road safety is a matter of utmost concern. With the growing population […]
ISO 37001 Certification: A framework for business operating system
Upholding the highest standards of integrity and ethical behavior is one of the most significant considerations for the businesses of […]
ISO 27001 Certification – Adhere to the best practices in information security
We all know that IT security, cybersecurity, and privacy protection are important for the companies to keep their standards safe. […]
ESG Reporting: A Strategic Imperative for sustainable investment for your business
ESG reporting has become one of the critical tools that companies have today, which reflects their commitment to sustainability, ethical […]
FSSC 22000 Certification: Your Commitment to food safety standards
Food is the first of the basic needs and a primary source of energy and existence. We relish it in […]
AS 9110 Certification: Needs and Benefits
Introduction to AS 9110 Certification A widely known and implemented quality management certification, AS 9110 stands as a specifically mastered, […]
R2 certification – Emphasize on the recovering of raw materials at the end of life
Responsible recycling – R2 certification What happens when you throw away your electronic materials like cellphones or computers? Have you […]
Boosting Facility Management in Saudi Through ISO 41001 Certification
Boosting Facility Management in Saudi Through ISO 41001 Certification The Ministry of Housing and Urban Affairs predicts that cities will […]
Transform Your Energy Management System With ISO 50001 certification
Searching for an organised approach to energy, a reduction in consumption and costs, a simpler system to manage and maximise […]
ISO 46001 Certification: Pioneering water efficiency for organizations
ISO 46001 Certification: Pioneering water efficiency for organizations With water shortages increasingly becoming a pressing issue, organizations across the globe […]
TISAX Certification in Saudi Arabia
Elevating Saudi Arabia’s Automotive Industry through Information Security with TISAX Certification In the era of hyper-connectivity, business information security is […]
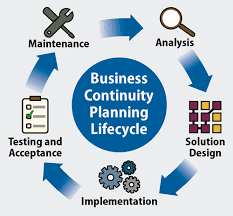
Important steps and information to implement ISO 22301 for your organization
Important steps and information to implement ISO 22301 for your organization The international business standard, ISO 22301:2019, acts as a […]
SOC Certification: Ensuring Security and Data Protection for Businesses
SOC Certification: Ensuring Security and Data Protection for Businesses System and Organization Controls (SOC) certification has emerged as a vital […]
SOC 1 and SOC 2 Attestation
SOC 1 Type and SOC 2 Type Explained All SOC types are audits using Organization Controls and American Institute of […]
PCI DSS Assessment Questionnaire
PCI DSS Assessment Questionnaire Most of the businesses these days accept credit cards and PCI DSS self-assessment questionnaire helps to […]
Difference Between ISO 14001 and RC 14001 and its Requirements
Difference Between ISO 14001 and RC 14001 “Broaden the scope of environmental management system” ISO 14001 and RC 14001-2023 are […]
Benefits of a PCI DSS
PCI DSS Assessment in Saudi Arabia PCI DSS which stands for The Payment Card IndustryData Security Standard is security standard […]
ISO 27701 Certification in Saudi Arabia
ISO 27701 PIMS Certification in Saudi Arabia A major concern in the world today is Data Privacy. This is because […]
BENEFITS OF HAVING A SOC ATTESTATION
SOC ATTESTATION System and Organizations Control attestations better known as SOC attestations is a certification and audit that evaluates the […]
What is the Importance of ISO 27001 Certification ?
ISO 27001 is a standard that is an information security management system that gives support to organizations thus establishing, implementing, […]
SOC Attestation Saudi Arabia
What is SOC ? Data security is a great menace in the world today. This comes in when technology is […]
FSSC 24000 Certificates: Ensuring Food Safety and Quality
Food safeties along with its quality are huge concerns for the consumers, regulators and the businesses in the same way. […]
Safeguarding Trust : Decoding System and Organizational Controls 1 (SOC 1) and System and Organizational Controls 12 (SOC 2 )Compliance for a secure digital realm
Safeguarding Trust : Decoding System and Organizational Controls 1 (SOC 1) and System and Organizational Controls 12 (SOC 2 )Compliance […]
ISO 50001:2018 : Energy Management System
One of the companies of Mohammed Abdulaziz Al-Rajhi and his sons, and it was founded by the late Sheikh Mohammed […]
ISO Saudi Assisted to Alshaya Group in obtaining ISO 45001:2018 Certification
Alshaya Group is one of the world’s leading brand franchise operators, offering an unparalleled choice of well-loved international brands to […]
ESG Sustainability Reporting
ESG Sustainability Reporting Environment Social Governanceor ESG as we know it encapsulates a holistic approach to business ethics and sustainability, […]
Helping Basha Medical Group Get ISO 13485 Certification
Basha Medical Group based in Riyadh, Kingdom of Saudi Arabia, is specialized in ophthalmic and ENT equipment and supplies. […]
ISO Saudi Assists JMBR Group in Obtaining ESG Certification
JMBR Group is a leading printing solutions provider in the Middle East with expertise in security printing. JMBR Group is […]
Ensure continued functionality with the SOC audit processes
“Reduce your cost of compliant upfront with us” If you are struggling to provide assurance around the risk management and […]
Infrastructure Application Security Assessment: Strengthening the Foundation of Digital Systems
In today’s interconnected world, the security of infrastructure applications is of paramount importance. Infrastructure application security assessments play a crucial […]
Android Application Security Assessment: Safeguarding Mobile Apps from Cyber Threats
As the popularity of Android applications continues to soar, ensuring the security of these apps has become more critical than […]
PCI DSS Assessment in Saudi Arabia
Enhancing Payment Security: In today’s digital age, protecting sensitive financial information has become paramount. Payment Card Industry Data Security Standard […]
National Cybersecurity Authority and CITC Communications and Information Technology Commission
ISO Saudi is a leading consulting firm in Kingdom of Saudi Arabia that offers a range of services to help […]
Validate your food safety management by getting the ISO 22000 Certification
Validate your food safety management by getting the ISO 22000 certification The ability of any certified organization to provide safe […]
ISO-SAUDI IS THE FIRST TO LAUNCH ISO 27001:2022 IN RIYADH, JEDDAH, SAUDI ARABIA
ISO-SAUDI IS THE FIRST TO LAUNCH ISO 27001:2022 IN RIYADH, JEDDAH, SAUDI ARABIA ISO 27001:2022 Security Controls – Annex A […]
ESG for Saudi
The Saudi Exchange as of late sent off its new ESG Disclosure Guidelines as a valuable asset to help giving […]
PCI DSS PAYMENT CARD INDUSTRY DATA SECURITY STANDARD CERTIFICATION
In the view of high end data breaches and hacking incidents, there are many reports for cyber security threats and […]
SOC 1 and 2 Type 1 and Type 2 Certification
SOC 1 and 2 Type 1 and Type 2 certification – Saudi Arabia That’s a huge record of compromised records […]
IITHRA – UNITED BUSINESS SERVICES – ISO 9001, 27001, 22301 CONSULTANCY AND CERTIFICATION
IITHRA – UNITED BUSINESS SERVICES – ISO 9001, 27001, 22301 CONSULTANCY AND CERTIFICATION ISO-Saudi is the First to Launch ISO […]
How SOC 2 attestation and certification helps people to manage data ?
How SOC 2 Attestation and Certification helps people to Manage data ? Privacy, security, availability, processing integrity, confidentiality…. “Data security […]
Benefits of ISO 45001 Certification
Benefits of ISO 45001 Certification Get ISO 45001 certification from ISO-SAUDI to benefit your business with worthy things. ISO 45001 […]
ISO-SAUDI Signs up with AFRAS for implementing ISO 9001 and ISO 41001 Facility management system
AFRAS X GQS It is generally said that Global associations trust Afras for its quality services which they give. It […]
WHAT ARE THE STEPS TO GET ISO 14001 CERTIFICATION IN SAUDI ?
What are the steps to Get ISO 14001 Certification in Saudi? Iso-Saudi contains an enormous, worldwide and multidisciplinary group of […]
What are the requirements of ISO 22000 Certification ?
What are the requirements of ISO 22000 certification ? ISO 22000:2018 Certification for Food Safety Management System is an interaction […]
Who can have ISO 27001 ?
Who can have ISO 27001 ? ISO-Saudi is the First to Launch ISO 27001:2022 in Riyadh, Jeddah, Saudi Arabia ISO […]
What is the process of getting ISO 27001 Certification in Saudi Arabia ?
WHAT IS ISO 27001 By ISO-SAUDI ? It is an international standard for Information Security Management Systems (ISMS) that provides […]
King Fahd University of Petroleum & Minerals – ISO-SAUDI signs up with King Fahd University for ISO 45001.
King Fahd University of Petroleum & Minerals – ISO-SAUDI signs up with King Fahd University for ISO 45001. King Fahd […]
ISO 27001:2022 – Information Security Management System
ISO-Saudi is the First to Launch ISO 27001:2022 in Riyadh, Jeddah, Saudi Arabia ISO 27001:2022 is a worldwide standard distributed […]
ISO 14001 Certification Benefits and its Process
ISO 14001 (Benefits& Process) ISO 14001 is a globally concurred standard that sets out the necessities for a natural administration […]
TERMS AND CONDITIONS, REFUND POLICY
Return Policy Since we are into services, there are no physical products which we offer to sell on our site […]
ISO Saudi signs up with Al Khaleej Books for implementing ISO 9001 Quality management system, ISO 14001 Environment management, ISO 27001 Info security management system
AI Khaleej Books ISO-Saudi is the First to Launch ISO 27001:2022 in Riyadh, Jeddah, Saudi Arabia ISO 9001 is a […]
ISO Saudi signs up with OPTERNA in Saudi Arabia
ISO Saudi signs up with OPTERNA in Saudi Arabia It is widely said that Global organizations trust OPTERNA products and […]
ISO Saudi signs up with MOSECO Jordan for implementing ISO 14001, ISO 45001, and ISO 9001 Integrated management system
ISO Saudi signs up with MOSECO Jordan for implementing ISO 14001, ISO 45001, and ISO 9001 Integrated management system An Integrated […]
ISO 41001 Certification Afras Trading Riyadh Saudi Arabia
ISO 41001 Certification Afras Trading Riyadh Saudi Arabia ISO 41001 is a worldwide administration framework standard that sets out the […]
AS 9100 / ISO 9001 certified SUMMIT HELICOPTER SAUDI ARABIA
TESTIMONIAL FROM THE MOST PREMIUM HELICOPTER COMPANY IN RIYADH, KINGDOM OF SAUDI ARABIA “Your Team was patient with me, and […]
QC 080000 – An Outline
QC080000 – An Outline Hazardous chemicals have become a major issue in the manufacturing industry, particularly in businesses producing toys, […]
What’s new in FSSC VERSION 6.0 ?
What’s new in FSSC VERSION 6.0 ? ISO-SAUDI in Saudi Arabia serves as the best ISO and FSSC certification providers […]
ISO 27001:2022 – Information Security Management Systems
ISO-Saudi is the First to Launch ISO 27001:2022 in Riyadh, Jeddah, Saudi Arabia ISO 27001:2022 accreditation was set up by […]
Dr SULAIMAN HABIB HOSPITAL GROUP @ FLOW MEDICAL IS CERTIFIED TO ISO 13485 MEDICAL DEVICE QUALITY MANAGEMENT SYSTEM MDQMS
We are glad to write a testimonial for ISOSAUDI who was instrumental in our organization getting (ISO 9001:2015 &IS013485:2016) certifications […]
CHANGES TO FSCC VERSION 6.0 Category C / I
CHANGES TO FSCC VERSION 6.0 Category C / I The new FSSC 22000 Version 6.0 was published on November 3, […]
INTRODUCTION OF ISO 41001:2018 (FACILITY MANAGEMENT)
INTRODUCTION OF ISO 41001:2018 (FACILITY MANAGEMENT) Facility management (FM) integrates a couple of disciplines so that you can have a […]
WHY CAN’T WE THROW THE ELECTRONIC DEVICES IN REGULAR DUSTBINS ?
WHY CAN’T WE THROW THE ELECTRONIC DEVICES IN REGULAR DUSTBINS ? Electronic devices are made up of heavy metals like […]
Catch up with the procedure of IFS certification
What is IFS? IFS are a safety standard that applies to companies that sell, supply, and manufacture food products. It […]
The Basics for implementing ISO 50001: 2018 – Energy Management System
The Basics for implementing ISO 50001: 2018 – Energy Management System The focus of all the leaders and the managers […]
Vulnerability Assessment and Penetration Testing
What is VAPT ? VAPT is a term used to describe security testing that is designed to identify and help […]
Purpose of ISO 13485 certification: Medical devices
Purpose of ISO 13485 certification: Medical devices In the medical industry, this ISO 13485 certification is made to be the […]
ISO 13485: Medical devices – Quality management systems requirements for Medical Devices
ISO 13485: Medical devices – Quality management systems requirements for regulatory purposes Raw materials Components Subassemblies Medical devices Sterilization services […]
ISO 13485 certification
FujiFilms Japan one of the largest conglomerates in Sale, Installation, Service of various types of Medical devices having global presence […]
Difference between management system certification and product certification: ISO 13485
Difference between management system certification and product certification: ISO 13485 If management conforms that they are meeting all the appropriate […]
The credibility of the ISO 13485 certification: Medical devices
Are you concerned about the credibility of the ISO 13485 certification? As medical professional or medical devices public procurement officer, […]
CE Certification for Technical Products
Ten Steps for CE Certification for Technical products by Notified body We provide complete solution for CE certification :- 1. […]
Data Sanitizing in SERI R2 Version 3
What is Data Sanitizing in SERI R2 Version 3 ? Many recycling vendors do have doubts and queries related to […]
ISO 13485 Medical Devices Certification
Dr Sulaiman Al Habib, Riyadh, Kingdom of Saudi Arabia – medical equipment groups signs up with ISO Saudi for training […]
AS 9145 – APQP For AS 9100 Aerospace
Many Aircraft Component Manufacturing Companies seldom overlook implementation of AS 9145 during AS 9100 Certification Interested in AS 9145 implementation […]
AS 9145 – Aerospace Standards for Advanced Product Quality Planning.
AS 9145 – Aerospace Standards for Advanced Product Quality Planning GQS supports in AS 9145 implementation, training and consultancy during […]
Environment Site Assessment as per ASTM E 1527
Green field projects requiring Phase 1 Environment Site Assessment as per ASTM E 1527 – Part 13 Be it Green […]
Electronic Recycling
Electronic Recycling Recycling of electronic products involve lot of responsible care with respect to Quality, Health and Safety, Ethical regulations […]
ISO 45001 Controls to Prevent COVID-19
ISO 45001 Controls to prevent COVID 19 Infections 1. Conduct HIRA 2. Consider COVID as a SignIficant Risk 3. Follow […]
GQS is Proud to Announce Certification Support Services in UK
GQS is proud to announce certification support services in UK. To know more info on certification in BRC Issue 8, […]
Proud To Announce The Launch of RBA Version 6 Consultancy
RBA Certification, RBA Consultancy, RBA consultant, RBA Responsible Business Alliance Certification in Riyadh, Dammam, Yanbu, Jeddah, Jubail, Hofuf . just […]
GQS Supports BOSCH in ISO 50001-2018, ISO 41001 Facility Management Certification
We are leading in consultancy and certification support to Large companies across various lines of biz both in India and […]
What is Anti Bribery Management System
ISO 37001 Consultant, ISO 37001 Certificate , ISO 37001 Consultancy, ISO 37001 Certification, ISO 19600 Certification, ISO 19600 Experts […]
Worlds First Trainings on ISO 22483
Worlds first Trainings on ISO 22483 auditor and general awareness. Courses offered include detailed insight into all the requirements of […]
ISO 22483 Launched for Hotels and Tourism.
First to launch ISO 22483 Certification for Saudi Hotels and tourism. Right from Gap analysis, Writing manuals, Procedures, Implementation support […]
Steps Of RBA Code of Conduct Certification
RBA Certification, RBA Consultancy, RBA consultant, RBA Responsible Business Alliance Certification in Riyadh, Dammam, Yanbu, Jeddah, Jubail, Hofuf just drop […]
Difference Between ISO 28000 & ISO 28001 Certification.
Supply chain security ISO 28000 experts with global experience in implementing in one of the largest warehouse in Asia […]
ISO 17024 Accreditation Requirements
ISO 17024 Experts for accreditation of training institutes which specifies criteria for the operation of a Personnel Certification Body […]
RBA Version 6.0
what is RLI, RMI, RFI in Responsible business alliance ? Want to learn more about labor, Mineral and […]
RBA Version 6
5 Pillars, 43 Requirements, 85 sub Requirements – RBA Responsible business alliance version 6 . What is AMA Auditee managed […]
World Standards Day
14th October 2020 – World Standards Day – SKY is the LIMIT Want to Learn more – email to [email protected]
RBA RESPONSIBLE BUSINESS ALLIANCE VERSION 6
Six route causes and its unique corrective action. Yes this is common in RCA, want to learn more about RBA […]
ISO 37001 ANTI BRIBERY MANAGEMENT SYSTEM
ISO 37001 Anti bribery management system certification by experts from management system. Right from training until implementation of the Whistle […]
ISO 13485:2016-Quality Management System
ISO 13485 Consultancy by IRCA Approved auditors with excellent experience in ISO 13485 audit experience in Medical devices such […]
ISO 46001 WATER EFFICIENCY MANAGEMENT CERTIFICATION
ISO 46001 Guidance for implementing in Manufacturing, Service, Process Industries by Industry expert with more than 25 years of […]
RAREST ISO INTEGRATIONS
Rarest combo in ISO Integrations.First in APAC to introduce with hands-on experience of implementing, training and certification with highest TAT […]
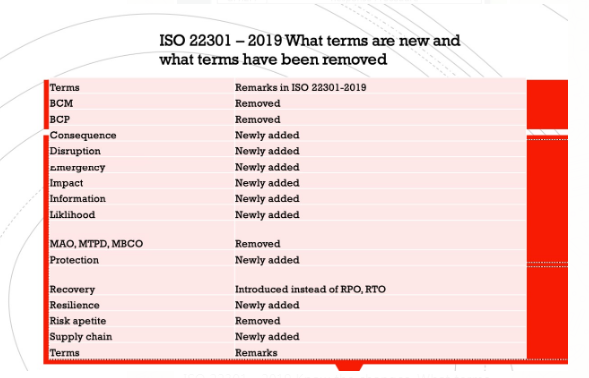
ISO 22301-2019 WHAT TERMS ARE NEW AND WHAT TERMS HAVE BEEN REMOVED
ISO 22301 – 2019 Know the changes. What terms are added in ISO 22301-2019 and what terms are revised […]
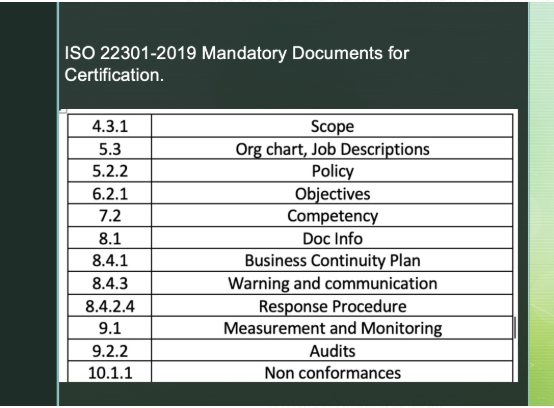
ISO 22301-2019 MANDATORY DOCUMENTS FOR CERTIFICATION
ISO 22301 Certification, ISO 22301 Consultancy, ISO 22301 consultant in Riyadh, Dammam, Yanbu, Jeddah, Jubail, Hofuf just drop an […]
RBA RESPONSIBLE BUSINESS ALLIANCE CERTIFICATION
RBA RESPONSIBLE BUSINESS ALLIANCE CERTIFICATION RBA Certification, RBA Consultancy, RBA consultant, RBA Responsible Business Alliance Certification in Riyadh, Dammam, Yanbu, […]
ISO-46001 WATER CONSUMPTION OUTLIERS DUE TO COVID-19
ISO-46001 WATER CONSUMPTION OUTLIERS DUE TO COVID-19
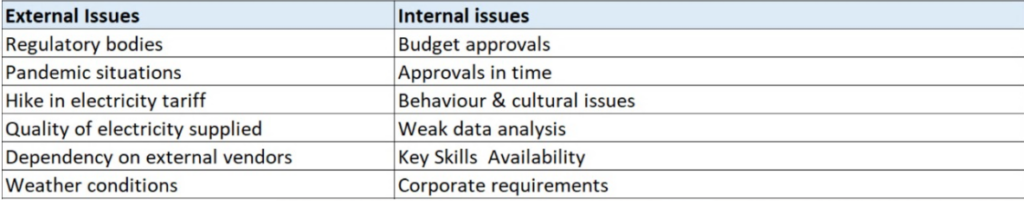
External & Internal Issues – ISO 50001-2018
Know the difference, call for queries regarding ISO 50001 Certification, ISO 50001 Consultancy, ISO 50001 consultant in in Riyadh, Dammam, […]
ISO 46001 WATER EFFICIENCY MANAGEMENT SYSTEM.
ISO 46001 WATER EFFICIENCY MANAGEMENT SYSTEM. Although this is the latest standard published we have carried out the highest number […]
Integrated management system IMS Certification Riyadh Jeddah Saudi Arabia
WHAT IS AN INTEGRATED MANAGEMENT SYSTEM? A management system that integrates all of organization system’s and processes into one single […]
Remote Online ISO Certification, online training due to Virus issue valid until December 2020
Remote Online ISO Certification, Online ISO General awareness Trainings – 1 day, 2 days Internal auditor training, 5 Lead auditor training […]
CTPAT, SCAN, ISO 28000 Certification in Saudi Logistic companies
ISO 28000:2007 CERTIFICATION in Saudi Logistic companies : Specifications for security management systems for supply chain Our world is a […]
ISO 13485 Certification in Riyadh, Jeddah Kingdom of Saudi Arabia
ISO 13485 CERTIFICATION SAUDI ARABIA – MEDICAL DEVICES QUALITY MANAGEMENT SYSTEM COMPLYING WITH SFDA SAUDI FOOD AND DRUG AUTHORITY […]
ISO 37001, ISO 26000, ISO 28000, SCAN, CTPAT Certification in Saudi Arabia
TIPS FOR EFFECTIVE AUDIT of ISO 37001, ISO 26000, ISO 28000, SCAN, CTPAT One of the mandatory requirements of any […]
12 STEPS INVOLVED IN CERTIFICATION ISO 27001- INFORMATION SECURITY MANAGEMENT SYSTEMS by SAUDI LOCAL AGENCY
Twelve 12 STEPS INVOLVED IN CERTIFICATION ISO 27001- INFORMATION SECURITY MANAGEMENT SYSTEMS by SAUDI LOCAL AGENCY Seder APD, Riyadh is […]
ISO 26000 compliance certification in Seder Riyadh Saudi Arabia
ISO 26000 compliance certification in Seder Riyadh Saudi Arabia ISO 26000 – Guidance on Social Responsibility and relation to ISO […]
ISO 28000 Supply chain security Riyadh Jeddah Rabigh
ISO 28000 Supply chain security Riyadh Jeddah Rabigh Be it SCAN Supplier Compliance Audit Network Compliance audit, Responsible business Alliance […]
2020 Best ISO 22301 Certification agency Saudi Arabia
ISO Saudi is a leading consultancy company in the Middle East. ISO- Saudi has employed top professional consultants who are […]
2020 Top ISO Certification Consultants Kingdom of Saudi Arabia
Who are the Top 3 ISO Certification consultants in Kingdom of Saudi Arabia for 2020 It is always important for […]
ISO 13485 Certification experts Saudi
ISO Saudi has recently signed a contract with ‘Leader Healthcare Group’ for the implementation of the ISO 13485:2016 standards for […]
Leader Group kick starts ISO 13485 Certification
Leader Group kick starts ISO 13485 Certification Seven Steps for ISO 13485 Certification in Jeddah at LEADER HEALTHCARE Conducting Risk […]
ISO 41001 Facility management Certification Riyadh Saudi Arabia
ISO 41001 Facility management system certification Comatec Saudi Arabia becomes the first company in Kingdom of Saudi Arabia to […]
AS 9100 Rev D Certification Jubail AASIA PRECISION
AASIA Jubail kick starts AS 9100 Revision D consultancy and certification project. AS 9100 Rev D for precision industries is […]
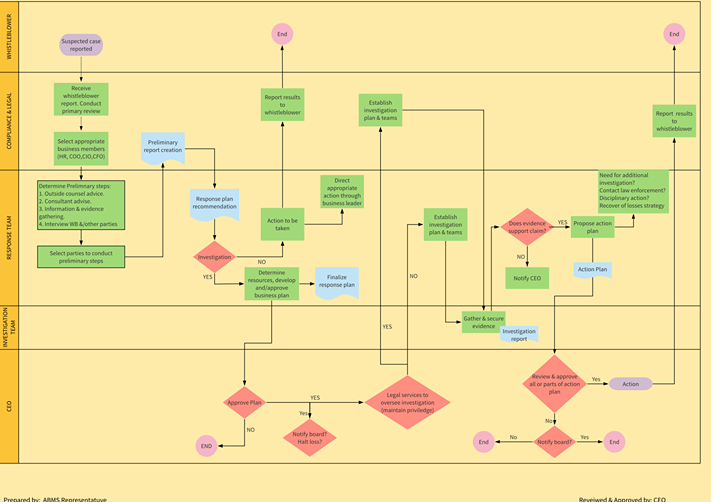
How to make a Business Continuity Management System effective?
Achievement of an ISO 22301 certification acts as a crucial component in an organization’s competitive edge so it becomes important […]
How does complying to ISO 22301 benefit your business?
An ISO 22301 is recognized globally and any industry implementing this standard will eventually pave the way to procure suppliers […]
Maintenance of a Business continuity management system.
The aim of the regular maintenance is to avoid an incident that could damage your reputation, affect your relationship with […]
Ensuring continual improvement in BCMS through ISO 22301 business continuity standards.
ISO 22301 takes a preemptive and a standardized stratagem to the improvement of the BCMS, through the recognition and execution […]
How to perform risk assessment or mitigation in your organization for ISO 22301
The purpose of risk management is to identify, reduce and recover from the impact of risk by devising risk management. […]
Setting the scope of your business continuity management system
ISO 22301 Business continuity management system provides a facility for an organization to choose to implement the standard on an […]
How to choose a certification body for ISO 22301 certification?
How to choose a certification body for ISO 22301 certification ? Apart from the price charged by the certification body […]
How to Leverage the benefits of internal audit in ISO 22301
How to Leverage the benefits of internal audit in ISO 22301 ? Recognition of nonconformities can begin from the identification […]
Top points required to be implemented for ISO 22301 certification.
Top points required to be implemented for ISO 22301 certification Setting up a management committee To look into the process […]
Resilience in business and improvement using ISO 22301 Certification
Resilience in business and improvement using ISO 22301 Certification ISO 22301 certification emphasises on the goal to make an organization […]
How to conduct a mock exercise in ISO 22301 business continuity management system.
Conducting a mock exercise gives some valuable business insights to identifies serious gaps. In order to prove that the organization […]
What is the importance of testing a business continuity management system?
What is the importance of testing a business continuity management system ? Testing a Business continuity management system is vital […]
How to form a strategic team for testing Business Continuity in ISO 22301
How to form a strategic team for testing Business Continuity in ISO 22301 A strong and expert team assigned to […]
Benefits of implementing a Plan Do Check Act Model ISO 22301
Benefits of implementing a Plan Do Check Act Model ISO 22301 The PDCA major role is to focus the organizational […]
Plan, Do, Check, Act and how it is applied to the Business Continuity
Plan, Do, Check, Act and how it is applied to the Business Continuity Let us understand the sequence Plan The […]
How to implement gap analysis in your BCMS effectively?
How to implement gap analysis in your BCMS effectively? A gap analysis is performed to check the loopholes in a […]
How is Business Impact Analysis and Risk Assessment related in a Business Continuity plan?
How is Business Impact Analysis and Risk Assessment related in a Business Continuity plan? ISO Saudi considers that there are […]
Why Choose ISO Saudi for an ISO 22301 Business Continuity Certification?
Our ISO Business Continuity Consultants hold expertise in the procedural implementation of the policies and strategy, making the organization resilient […]
The process to get ISO 22301 certification with the help of ISO Saudi
The process to get ISO 22301 certification with the help of ISO Saudi Choose ISO Saudi to get an ISO […]
How can an organization get an ISO 22301 Business Continuity Management System Certification?
How can an organization get an ISO 22301 Business Continuity Management System Certification? ISO Saudi can help you get an […]
Benefits of Business Continuity Management System and its Certification
The prime need of a BCM system is to ensure continuity in business functions, maintaining service agreements with customers by […]
What organizations should implement ISO 22301: Business Continuity Management System?
What organizations should implement ISO 22301: Business Continuity Management System ? It is most important for any business to function […]
BCM and the steps to implement ISO 22301
BCM and the steps to implement ISO 22301 As outlined in the ISO 22031:2012 standard, BCM is defined as a […]
Implement ISO 22301 Business Continuity Management System?
Implement ISO 22301 Business Continuity Management System ISO 22301 specifies in its standard that organizations must understand its requirements. Business […]
What does ISO 22301 certification mean ?
What is ISO 22301 Certification mean? Eventualities in business cannot be predicted and if occurred can bring a huge loss […]
ISO 13485 Communications Best Practices
Consultants over the world agree on the fact that proper communication is one of the key requirements of the ISO […]
Top Advantages Of ISO 13485 Based QMS
It may often become difficult for consultants to explain the benefits of the ISO 13485 certification to company executives. In […]
ISO 13485 Product Design Issues
Among the many requirements of the ISO 13485 certification as required by the Saudi Food and Drug Authority, product design […]
Why Spreading Awareness For ISO 13485 Is Important
The Saudi Food and Drug Association (SFDA) urges all medical device manufacturers to get the ISO 13485 certification. ISO 13485 […]
Factors Which Determine ISO 13485 Calibration Frequency
Calibration of medical devices is an issue given prime importance in the ISO 13485 certification standard. The SFDA, along with […]
Four Reasons Product Owners Should Get ISO 13485 Certification
Despite repeated insistence from the Saudi Food and Drug authority for getting the ISO 13485 Certification, medical product manufacturers may […]
How To Engage Top Management In ISO 13485 QMS
In order to ensure the proper execution of a QMS under the ISO 13485 certification, the Saudi Food and Drug […]
Advantages Of ISO 13485 Calibrations
We have already discussed about what calibration is according to the ISO 13485 certification and why it is important. In […]
Top ISO 13485 Tips For Measuring Product Performance
The responsibility of a medical device manufacturer in Saudi Arabia does not end with the release of the device. According […]
Top Tips For ISO 13485 Calibration
Calibration being one of the significant requirements for ISO 13485 according to the Saudi Food and Drug Authority, consultants stress […]
What Are ISO 13485 Calibrations?
One of the significant requirements in the ISO 13485 certification standard is the need for proper calibration of all medical […]
General QMS Requirements For ISO 13485
For medical device manufacturers in Saudi Arabia, the ISO 13485 certification is one of the required Quality Management System (QMS) […]
Top Training Requirements For ISO 13485
As per the requirements of the ISO 13485 certification, all involved personnel must have the requisite training, skills and competency […]
How To Choose The Right ISO 13485 Consultant
One of the essential requirements for any company in Saudi Arabia to achieve the ISO 13485 certification is to choose […]
Top Mistakes With ISO 13485 Documentation Control
Proper documentation is one of the essential prerequisites for ISO 13485 certification. Yet, according to most consultants this is where […]
Top Certification Tips For ISO 13485
ISO 13485 being one of the most important certifications for medical device manufacturers, most businesses look towards expert consultants in […]
ISO 13485: A Few FAQs
The Saudi Food And Drug Authority recommends ISO 13485 as one of the required certifications for medical device manufacturers. Today […]
Top Hurdles In ISO 13485
Getting the ISO 13485 certification required by the SFDA standards is one of the prerequisites for medical device manufacturers in […]
Do You Need A Consultant For ISO 13485 Certification?
One of the questions that come to the mind of every medical device manufacturer in Saudi Arabia is whether to […]
How To Create A Medical Device File MDF In ISO 13485?
Consultants advice medical device manufacturers in Saudi Arabia to prepare for ISO 13485 certification by creating a medical device file […]
How ISO 13485 Can Improve Business
Medical device manufacturers in Saudi Arabia often make the mistake of underestimating the importance of ISO 13485 certification. Though recommended […]
Similarities And Differences Between ISO 13485 And Other ISO Certifications
ISO 13485 certification is one of the recommended certificates that medical device manufacturers in Saudi Arabia are advised to get […]
ISO 13485: Things To Know
ISO 13485 is an essential certification that all medical implement suppliers need to get as per the Saudi Food And […]
How 13485 Certification Can Help Medical Device Manufacturers In Saudi Arabia
The Saudi Food And Drug Authority encourages all medical device manufacturers in Saudi Arabia to go for ISO 13485 certification. […]
Hyundai car Genesis Car Distributor Wallan Trading Riyadh ISO Certification
Wallan Trading a largest Hyundai Genesis car distributor in the Middle east has signed up with ISO Saudi for certification […]
Why Use ISO 13485
The ISO 13485 certificate is meant for assessing the quality requirements for the design and manufacture of medical devices. This […]
What is ISO 22301 certification in Riyadh ?
Eventualities in business cannot be predicted and if occurred can bring a huge loss and disrupt the normal functioning of […]
Check if your ISO Auditor is Valid
Check if your company is offering certification in 1-7 days. Why ? Most companies require at least 2-3 months for […]
Steps for ISO 13485 SFDA Certification
Steps for ISO 13485 SFDA Certification ISO Saudi prepares medical device manufacturers and distributors for ISO 13485 Certification as per […]
Why ISO 13485 certificate is important for Medical Distributors in Saudi
Why ISO 13485 certificate is important for Medical Distributors For ISO 13485 certification requirements firstly SFDA Saudi Food Drug Authority […]
Free download ISO 13485 pdf audit checklist, ISO 14971 pdf Riyadh Jeddah
Free download ISO 13485 pdf audit checklist and ISO 14971 pdf If you wish to download a free copy of […]
ISO 13485 SFDA Requirements Certification Riyadh, Jeddah
ISO 13485 SFDA Requirements Certification in Riyadh, Jeddah Few of the key features and requirements of the SFDA Saudi Food […]
SFDA ISO 13485 Certification
Medical device companies to be ISO 13485 certified in Saudi Arabia ISO 13485 certification involves preparation of the 1 . […]
ISO 18788 Certification – Security Service in Riyadh, Jeddah, and Dammam
Strengthen Your Security Operations with ISO 18788 Certification Security services are a necessity for establishing trust, safety, and order across […]
Free Download ISO 22301-2019, ISO 45001-2018, ISO 22000-2018, ISO 50001-2018-pdf
Yes free advice to download the ISO 22301-2019 – Business continuity management system including the 4 steps required for Business […]
ALNAHDI Aviation Jeddah AS 9100, ISO 9001 Certification
AS 9100 Certification, ISO 9001 Certification Excellence is the core value of Alnahdi United for 60 years. From the family […]
TAMS Aviation Riyadh is now certified to ISO 9001-2015
TTS, Riyadh Kingdom of Saudi Arabia is a Premier Solutions Provider in the areas of procurement & logistics, technical training […]
SERI – R2 Responsible recycling launched in Saudi Arabia, Muscat, Dubai
ISO Saudi becomes the first to launch the R2 Responsible recycling standards in Riyadh, Jeddah, Muscat and Dubai. The standards […]
R2 SERI Certification Riyadh Jeddah Dubai Muscat Kuwait
Yes you heard it right..ISO Saudi becomes the first in Gulf region to introduce Responsible recycling R2 SERI approved certifications. […]
Seder Group starts ISO 27001, ISO 26000 Certification
Seder AMD a part of the large SEDER group has kick started the ISO 27001 Information security and implementation of […]
Saudi Arabian Cement manufacturers ISO 22301 Certification
ISO 22301 in manufacturing companies,ISO 22301 Certification, ISO 22301 Consultant, ISO 22301 Certificate Business Continuity management system can be implemented […]
Al Hamrani Group starts ISO 22301 Business Continuity Management
Al Hamrani Group a leading group in Kingdom of Saudi Arabia, has kick started with ISO Saudi for implementing the […]
GCC Secretariat Riyadh ISO 9001:2015 Certified
Gulf Cooperation Society Secretariat with its Head office in Riyadh Kingdom of Saudi Arabia has been certified to ISO 9001:2015. The […]
ISO 41001 Certification for Facility management
10 Steps for ISO 41001 Facility management certification Document the Context of the organisation Document the Needs and expectations of […]
ISO 20658 Certification for Medical Transport Company, Riyadh Saudi Arabia
MTC Riyadh achieved the ISO 20658 certification which is one of it kind in Saudi Arabia. This certification process […]
ISO 9001:2015 Certification started in Seder Head Office
ISO 9001:2015 Certification support to SEDER Head Office has been initiated. Covering departments such as IT, HR, Procurement, Admin, Finance […]
AS 9100, AS 9110 Certification Riyadh, Jeddah
Want to implement AS 9100, AS 9110 latest version in Riyadh, Jeddah, ensure that the following are documented for easy […]
ISO 50001:2018 Launched Riyadh Saudi Arabia, Free download.pdf
What are the changes, what does an organisation need to carry out to be certified to ISO 50001-2018 new version, […]
ISO 45001-2018 Certificate Riyadh Saudi Arabia
Alfaa Metal Coating ( AMC ) is a leading provider for various metal coating services in the UAE. Alfaa has […]
Adwan chemicals Riyadh ISO 45001-2018, ISO 14001-2015, ISO 9001-2015 Certified
Adwan chemicals the largest silica based chemicals manufacturing company in the Gulf. The company has now certified to ISO 45001-2018, […]
ISO 9001-2015, ISO 45001-2018, ISO 14001-2015 Certificate Telecom Saudi
Bazy Trading and Contracting Co a company based in Riyadh , Kingdom of Saudi Arabia is a leading provider of […]
ISO 13485 Medical Devices Consultants
ISO 13485 Medical Devices Consultants ISO 13485 standard convenes with the essentials of quality management systems with reverence to the […]
ISO 22301 Business Continuity Consultants
ISO 22301 Business Continuity Consultants It’s almost unfeasible to plan for every disruptive event that could occur, so we use […]
ISO 22301 Business Continuity Certification
ISO 22301 Business Continuity Consultancy (ISO 22301 – Business Continuity Certification) ISO 22301 Business Continuity Consultancy, Certification presents parcel and […]
ISO 13485 Medical Devices Consultancy/Services
ISO 13485 Medical Devices Consultancy/Services Medical Devices are extremely regulated products & different nations have diverse regulatory tactics for market […]
ISO 13485 Certification for Medical Devices
ISO 13485 Medical Devices Certification (ISO 13485 Certification for Medical Devices) Evolving in the Medical Device field calls for a […]
ISO 22301 Certification
ISO 22301 Business Continuity Certification (ISO 22301 Business Continuity Management System Certification) ISO 22301 Business continuity management (BCM) is the […]
ISO 22301 Business Continuity Certificate
ISO 22301 Business Continuity Certificate ISO 22301 is the Business Continuity Certificate System set. ISO 22301 Certificate has been developed […]
ISO 13485 Medical Devices Certification Riyadh
ISO 13485 Medical Devices Certification (ISO 13485 Certification for Medical Devices) Being ISO 13485 certified helps to combine business and […]
ISO 22301 Business Continuity Management Certification
ISO 22301 Business Continuity Management Certification ISO 22301 opens out the necessities for a best-practice business continuity management system (BCMS). […]
ISO 13485 Medical Devices Certification
ISO 13485 Medical Devices Certification – Quality Management Systems The ISO 13485 standard is a viable solution for meeting the […]
FSSC 22000 – V4.1 – Do you know ?
FSSC 22000 – V4.1 Certification- Do you know ? Do you know the actual requirements and the time line from […]
Upgradation to ISO 22000-2018 certificate – Food safety management system
ISO 22000-2018 Certificate, ISO 22000-2018 Consultants, ISO 22000-2018 Consultancy, ISO 22000:2018 Certificate, ISO 22000:2018 Upgrade Yes, with the recent publications […]
ISO 22301, ISO 27001 Banks, ATM, Certification consultants Saudi Arabia Riyadh
ISO 22301, ISO 27001 Banks, ATM, Certification consultants Saudi Arabia Riyadh With more and more hacking attempts by unknown sources […]
Challenging ISO 9001:2015 Certification in Riyadh
Are ISO requirements challenging for implementation ? Contracting to an ISO consultancy firm would imply that you set up quality administration framework […]
ISO 14001-2015 Certification Riyadh, Jeddah, Yanbu
ISO 14001-2015 Certification Riyadh, Jeddah, Yanbu As we are as of now seeing the sudden change in the atmosphere because […]
ISO 22301 Certificate – First Powder company in Riyadh Saudi Arabia
NAFCO Riyadh becomes the first powder coating company in Riyadh to be certified to ISO 22301. Did you ever think […]
Five Steps for ISO 22301 certification – Riyadh Kingdom of Saudi Arabia
FIVE STEPS FOR ISO 22301 CERTIFICATION Plan and conduct Business Impact assessment Add Key staff Start Risk assessment Identify RTO […]
ISO 22301 Certification Consultant Riyadh, Jeddah Saudi Arabia
FIVE IMPORTANT STEPS REQUIRED FOR ISO 22301 BUSINESS CONTINUITY CERTIFICATION For all companies the need of the hour either during […]
Kayes Riyadh starts ISO 9001:2015 Certification process
KAYES Trading company kickstarts their ISO 9001-2015 Certification program. The gap analysis including PESTL Analysis, SWOT Analysis was carried out […]
AS-9100-D-2016-Certification
ISO SAUDI was approached by SRSC ( Saudi Rotorcraft Support Company) for training it’s personnel on AS 9100 Rev D. […]
Kanoo Group signs for ISO 9001:2015, ISO 14001:2015
BDP Kanoo is a part of Kanoo group and is based in Dammam. The company is into logistics and freight […]
ISO 9001:2015, ISO 14001:2015, OHSAS 18001:2007 Certificate Algiers Algeria
Adwan Chemical Industries Company having its branches in Riyadh, Dammam and Algiers Algeria had expressed its interest in implementing the […]
ISO 9001-2015 Certification, ISO 9001-2015 Consultants
ISO Saudi successfully completes its training programs on ISO 9001-2015 auditing covering ISO 9001-2015 Understanding the organization ISO 9001-2015 Identifying […]
GACA Auditing ISO 9001:2015 Auditing Riyadh
Infinity Aviation Academy one of the largest and the best aviation academy in Saudi Arabia completes the ISO 9001:2015 Auditing training […]
ISO 9001:2015 recommended for Ministry of Health
Ministry of Health ARAR, is now certified for ISO 9001:2015. The medical supply warehouse consisting of various warehouses covering all […]
Almoosa specialist hospital – kitchen is HACCP certified
It was really very useful tool to improve the level of service in our dietary department . it is for […]
Al Hamrani Jeddah Testimonial ISO 9001:2015
We are extremely happy with the services provided by ISO-SAUDI to achieve the ISO 9001:2015 Certification. The complete project was […]
JCI Accreditation, JCI Consultants, JCI Certification for Hospitals in Saudi Arabia
JCI Accreditation, JCI Consultants, JCI Certification for Hospitals in Saudi Arabia Steps involved Expert visit to assess the processes Gap […]
UKAS ISO 9001-2015, UKAS ISO 14001-2015, UKAS OHSAS 18001
ISO Saudi offers UKAS ISO 9001-2015, UKAS ISO 14001-2015, UKAS OHSAS 18001 certification to any kind of companies in UAE, Kuwait, […]
AS 9100 Rev D Transition AS 9100 Revision D Consultant, AS 9100 D 2016 Certificate
AS 9100 Rev D Transition AS 9100 Revision D Consultant, AS 9100 D 2016 Certificate AS 9100 Aerospace standards Revision […]
ISO 9001-2015 Training, ISO 9001-2015 Certificate
Al Harmani Group Jeddah Completes the Auditors training program in ISO 9001:2015 Quality Management System. The training consisting of ISO […]
Saudi Master Baker All units ISO 22000 by SGS Saudi Arabia
ISO 22000 Consultancy, ISO 22000 Consultant, ISO 22000 Certificate Saudi Masterbaker Dammam, Jeddah and Riyadh all Five plants are now […]
Alhamrani Group – Universal company Jeddah ISO 9001:2015 Certificate
Alhamrani Group – Universal company Jeddah which deals with Diebolt ATMs Sales, Service and Spares all through KSA, Dubai has […]
Bolan Steel, Riyadh is ISO 9001 Certified
BOLAN COMPANY FOR STEEL in Riyadh is now certified to ISO 9001. The team involved in complete implementation since 4 […]
AL HALEEF Group Paint company is now certified to ISO 9001
Al Haleef Group for Paints and Colored dust has successfully implemented the ISO 9001 Quality management system standards. The company […]